SLU
EAS-A193 Class Notes
Earth's Origin and
Composition
Composition
|
SLU EAS-A193 Class Notes |
Composition |
In astronomy and cosmology, distance is often represented using the units called "light-years". A light-year is the distance that light travels in one year, and since light travels at approximately 3x10^8 meters/second and there are approximately 3.15x10^7 seconds in one year, a light-year (distance is the velocity multiplied by the time) is just under 10^16 (10,000,000,000,000,000) meters, or about 10 trillion kilometers.
The distance from the Sun to Pluto, which is the dimension of our solar system, is about 5.4 light-hours and the distance to the nearest star, Proxima Centauri, is about 4.25 light-years. Our solar system is part of the Milky Way Galaxy, which is a typical size galaxy and is about 100,000 light-years across. Andromeda, our nearest galaxial neighbor, is located about 2,140,000 light-years away and the distance to the farthest object we can see is about of 10-20 billion light-years.
December opens with an increase in oxygen in our atmosphere, a by product of oxygen-producing algae that destroy themselves by overproducing oxygen. The first worms appear in mid-December, and plants begin colonizing the land around December 20. Life enters middle age on Christmas and dinosaurs dominate the next few days. The first primates appear on the December 29th but the first humans don't develop until late evening on the last day of the year, near 10:30 PM. Agriculture is invented with just 40 seconds before midnight, Rome falls with three seconds remaining, and recent history makes up the last second. We are beginning the second year as newcomers to the universe.
Our current, best hypothesis for the origin of the universe is called the Big Bang. The cause of the Big Bang is unknown, and so is what, if anything, went on before it. But we have learned, and are continuing to learn much about how the universe evolved since its birth. The existence of a universe-initiating explosion is supported by several lines of evidence, most notably the observed motions of the galaxies, the observed background electromagnetic hiss generated during the bang, and evidence from the observed abundance of light chemical elements in the universe.
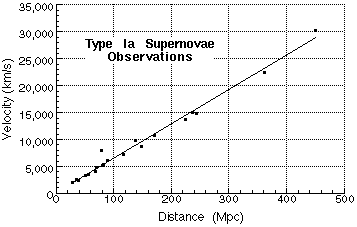
Observed velocities of distant supernovae measured in 1996 by Adam Reise, W. H. Press, and R. P. Kirshner. Hubble's original data only included measurements from a few Mpc distance (Mpc represents megaparsec which is a distance equivalent to 3.24x10^6 light-years).
The slope of the line in the Hubble diagram is 63.6 kilometers per second per megaparsec (a megaparsec is 3.26 million light-years)
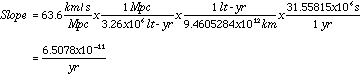
The inverse of the slope of the line in the diagram is called the Hubble age of the universe, which in this case is 15.4 billion years, near the center of the typical range of universal age estimates.
Everything we know of is a composite of a mere 109 building blocks that we call elements. Atoms of the 92 naturally occurring elements combine to form the myriad of materials that we see and use every day. An atom is a collection of particles called protons, neutrons, and electrons (these particles are composites of still smaller particles, but we'll keep it simple). We can imagine an atom as a dense core of protons and neutrons called the nucleus, surrounded by a cloud of orbiting electrons and since protons and neutrons are much more massive than electrons, most of the mass of an atom resides in the nucleus.
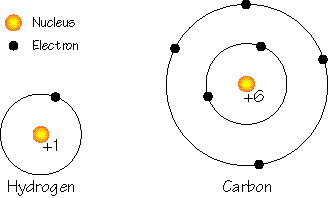
Atoms are electrically balanced objects with a nucleus of protons and neutrons surrounded by a cloud of moving electrons. The charge of the nucleus, or the number of protons in the atom, uniquely identify each element. Occasionally an atom may gain or lose an electron creating a charge imbalance and producing what we call an ion.
The number of neutrons in the nucleus can vary and we call atoms of one element that have different numbers of neutrons, isotopes of that element.
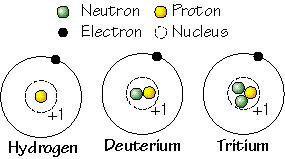
The number of neutrons in an atom may also vary, producing what we call isotopes. Some isotopes of certain elements are unstable and "decay" into other isotopes or elements. We'll discuss below how these important transitions produce heat and provide natural clocks.
The periodic table is a scientific tool that helps us understand the behavior of materials. In the table, elements are ordered by the number of protons (or electrons), which we call an element's atomic number, and arranged into columns which group elements that display similar properties. Click here to link to a site dedicated to periodic table.
All matter in the universe was generated during the Big Bang but much of it has been reworked in the interior of stars.
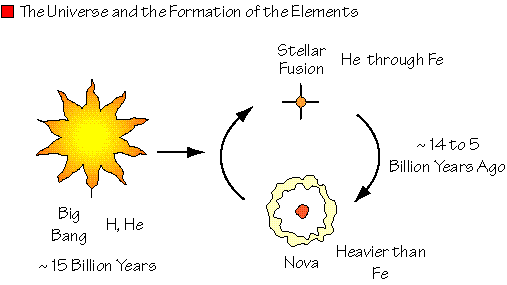
Light elements were formed during the early stages of the big bang, creating a universe composed principally of hydrogen and helium. Since then, the heavier elements are manufactured by fusion processes within stars or during spectacular supernova explosions.
Our understanding of the processes that formed the solar system comes from our understanding of the physics of rotating bodies and investigation into the chemical composition of the Sun, planets, moons, and asteroids. Although much exciting work remains to be done on many aspects of solar-system evolution, we have a robust hypothesis describing the solar-system formation that provides a stable framework for research.
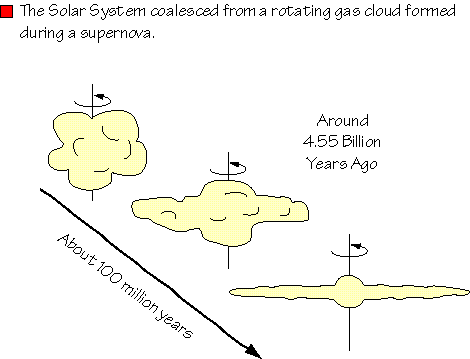
The solar system formed when a rotating gas cloud, the result of at least one prior supernova, was perturbed by a passing object (or another supernova) and began to collapse as a result of gravitational attraction.
Types of Meteorites
Methods for dating the age of rocks and meteorites exist&endash; they are based on the spontaneous transition of certain elements to other elements and called radiometric dating techniques. The spontaneous transition is called radioactive decay and we call the isotope that decays the "parent" and the product of decay a "daughter".
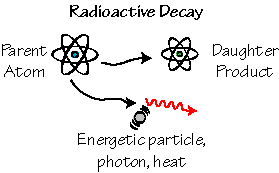
Radioactive decay one atom spontaneously changes to another, releasing heat, light, or another atomic particle. The original atom is called the "parent", the new atom is called the "daughter".
The result of such analyses indicate that meteorites have ages near 4.56 Ga, and hence so does Earth. The oldest rocks are just under 4 Ga, no rocks survived the earliest part of Earth history.
From spectral analyses of the sun and comparison with meteorites, we can estimate the abundance of elements in the solar system.
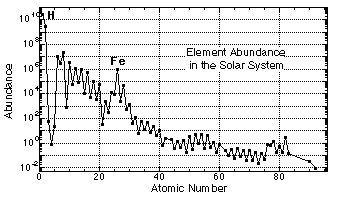
Solar-system abundances estimated from solar photosphere spectra and chondritic meteorite compositions.
After inferring the original composition of the solar system from the sun and meteorites we can fill in gaps in our knowledge of Earth's composition.
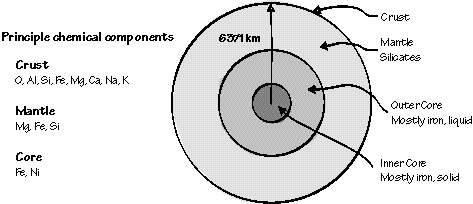
Structure and major chemical composition of Earth.
Earth's radius is about 6,371 km and the radius of the core is about 3,486 km (the inner core radius is about 1,217 km&endash; a little more than two-thirds of the radius of the Moon). Earth's mass is approximately 5.973x1024 kg, and its mean density is 5.515 g/cm^3. The typical density of continental rocks is about 2.7 g/cm^3. crust accounts for less than one-half of one percent of the mass of the planet. The mantle accounts for about 84% of Earth's volume but the core contains almost 70% of the planet's mass.
Earth is hot because of three heat sources
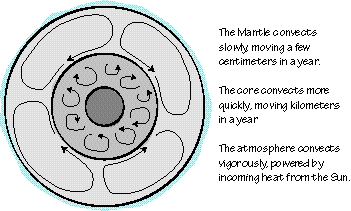
Major convective systems in the solid part of Earth. The core convects at a rate of 10 km/yr, the mantle is much more sluggish moving along at a rate of about 10 centimeters per year.
The motions deep in Earth are much slower than those common at the surface. Motion in the outer core proceeds as a rate on the order of ten km/hr (about six miles per hour). This may seem slow, but the material is under enormous pressure and that slow motion is the source of Earth's magnetic field. The mantle is much more sluggish, creeping along at a rate of about ten centimeters per year, about 100,000 times slower than the core. But these slow moving processes have had a tremendous impact on the character of our planet.
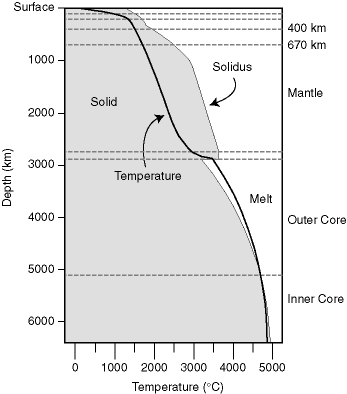
Allegre, C., From Stone to Star, A View of Modern Geology, Harvard University Press, 1992, 287 pages.
Atkins, P. W. The Periodic Kingdom, A Journey into the Land of the Chemical Elements, Science Masters Series, Basic Books, a Division of Harper Collins Publishers, 1995.
Barrow, J. D, The Origin of the Universe, Science Masters Series, Basic Books, a Division of Harper Collins Publishers , 1994, 150 pages.
Dalrymple, G. B., The Age of the Earth, Stanford University Press, 1991,474 pages.
Ferris, T. , The Whole Shebang - A State of the Universe(s) Report, Simon & Schuster, 1997, 393 pages.